RESEARCH
MESOSCALE & MOLECULAR TAU BIOPHYSICS
 | Our research seeks to uncover the physical principles that govern how tau proteins condense, transform, and misfold into pathological aggregates. We develop mechanochemical models that couple tau phosphorylation and hyperphosphorylation dynamics with intermolecular interactions and ionic environments to explain how normal condensates evolve into fibrillar tangles seen in CTE and related tauopathies. By integrating quantitative modeling with collaborative experimental validation, we aim to reveal how non-equilibrium biochemical regulation drives mesoscale organization and pathological transitions. This framework aspires to identify new physical biomarkers and intervention strategies targeting early stages of neurodegeneration. |
CELL SCALE CYTOSKELETAL MECHANICS
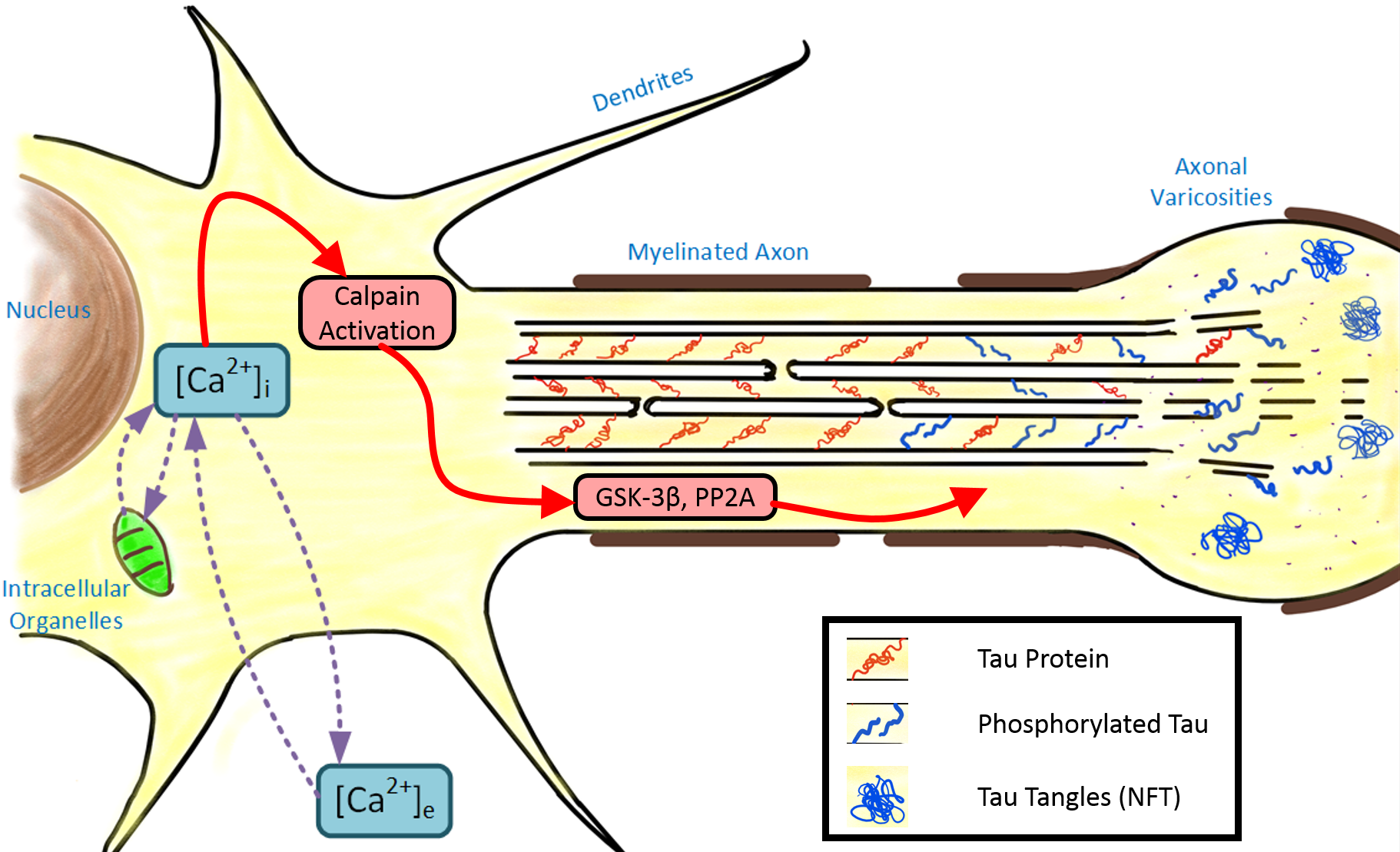 | We investigate how tau phosphorylation dynamics orchestrate the self-organization of the axonal cytoskeleton. Building on our models of tau phase separation, we explore how optimally phosphorylated tau drives microtubule growth, alignment, and cross-linking to form mechanically resilient axonal bundles. By coupling biochemical regulation with cytoskeletal force transmission, our work bridges molecular kinetics and cellular mechanics to reveal how structure and stability emerge from active processes. Extending this framework, we aim to uncover how hyperphosphorylation and injury disrupt cytoskeletal architecture, offering new insights into the mechanical fragility and progressive degeneration of axons following traumatic brain injury. |
TISSUE SCALE BRAIN INJURY SIMULATIONS
 | Our long-term goal is to bridge cellular pathology with brain-scale mechanics to better understand the progression of traumatic brain injury (TBI) and chronic traumatic encephalopathy (CTE). While current TBI models largely focus on stress and strain distribution, we seek to integrate the secondary biochemical and neurodegenerative effects emerging from tau dysregulation and cytoskeletal disruption. By coupling multi-scale mechanochemical models—from axonal dynamics to tissue-level deformation—we aim to uncover how local cellular injury cascades into global brain dysfunction. This framework aspires to redefine predictive modeling of brain injury by incorporating the feedback between neurons, glial cells, and the surrounding tissue microenvironment. |